Developing New Treatments for Pain
One in five Australians lives with chronic pain, a debilitating disorder which also afflicts adolescents and children, and this rises to one in three people over the age of 65. Chronic pain is characterised by the sensation of physical pain in the absence of noxious external stimuli. This project aims to identify and develop new molecules that could be used to study and treat chronic pain
Area of science
Biological Sciences, Biophysics, Chemical Sciences, Geosciences
Systems used
Magnus
Applications used
GROMACSThe Challenge
Chronic pain is a multifaceted disease involving dysfunction of a number of proteins, such as the acetylcholine receptors, and so-called “ion channels”- proteins which are responsible for transmitting signals in the nervous system. How the dysfunction of these proteins affect the development and progression of chronic pain needs to be understood. Moreover, new molecules which can either inhibit, or activate, specific nervous system proteins are needed to develop effective treatments
The Solution
High performance computing (HPC) technology can be used to study how proteins and molecules move, and provide insights into the ways in which new molecules can bind to target proteins in the nervous system. With this information, new molecules can be designed which can bind more strongly to their target proteins, with greater specificity. These new molecules can be developed into drugs in future
The Outcome
HPC facilities at the Pawsey Centre enable the movements of proteins and molecules to be studied over a sufficiently long time to ensure that the simulations are accurate, and that the predicted protein movements and their binding mechanisms to various molecules are realistically represented
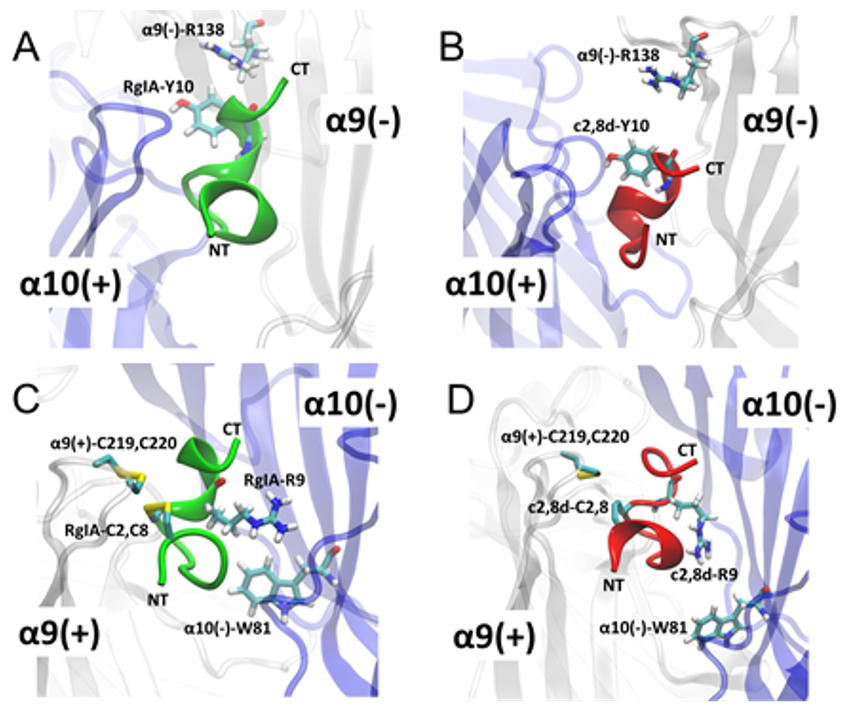
Figure 1. Key interactions between native and chemically-modified RgIA toxin with a human acetylcholine receptor subtype