Energies and Adsorption in Complex Nanoporous Materials
The project aims to understand the stabilisation of surface coatings and adsorption of molecules and ions at surfaces Supercapacitors are a class of energy storage that relies on adsorption of ions at electrode surfaces. We investigate the role that quantum mechanical properties of ions have on adsorption. The aim is to identify why some electrolyte solutions provide better energy storage performance than others. A related question is the effect of saline solution on the behaviour of polymer films, including synthetic coatings and biological membranes. The aggregation of protein molecules, or adhesion to surfaces, varying with saline composition and can be controlled or optimised by selection of salt placed in solution. The same physics affects the release of greenhouse gases trapped in ice (gas hydrates), with quantum mechanical interactions influencing the rate at which gas molecules may be released from melting ice.
Area of science
Chemical Sciences, Chemistry, Colloid and Surface Chemistry, Geosciences
Systems used
Magnus
Applications used
nwchem, FEniCS, lammpsThe Challenge
The are two parts to the challenge. Firstly, we needs a means of describing the forces originating from quantum mechanics which are experienced by ions and molecules. These influence adsorption of ions onto surfaces.
Then, we need means of evaluating the overall impact that ion adsorption has on the energy storage characteristics of supercapacitors, or on the adhesive forces between protein molecules or other microparticles
The Solution
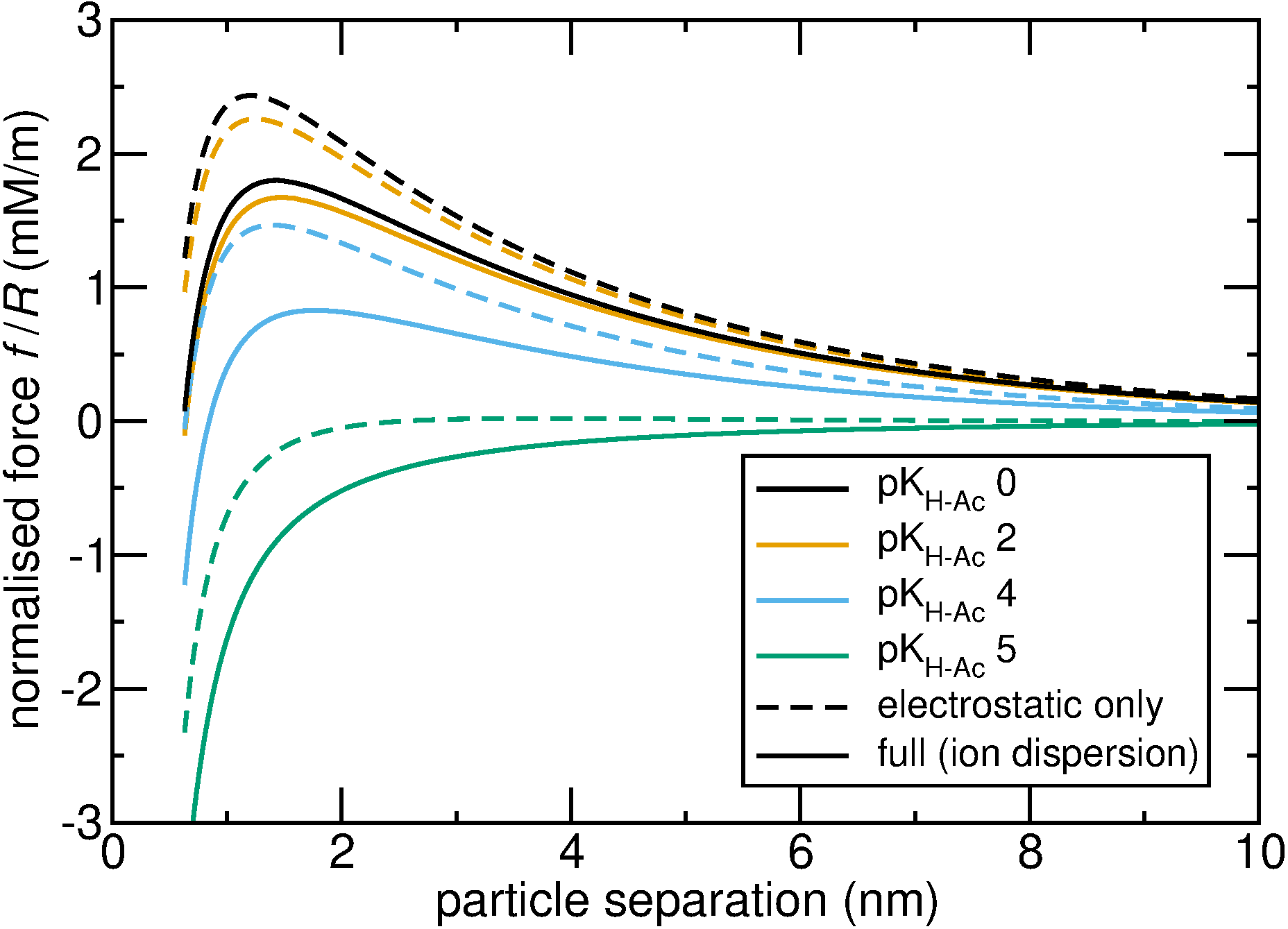
The theory of van der Waals forces provides a means of evaluating quantum mechanical interactions with large-scale surfaces. In the case of ions, the theory centres on the polarisability of the ion, which is a quantum mechanical property that quantifies how readily the electron cloud of the ion can move in response to electric fields.
Once the van der Waals interaction has been evaluated with the aid of quantum mechanical polarisabilities, the ensuing ion adsorption layers and resultant electric fields can be calculated by application of Poisson-Boltzmann models. Finite-element methods provide an effective means of computing solutions for Poisson-Boltzmann equations.
The Outcome
Pawsey Centre resources enable deployment of NWChem, quantum chemical software which has been designed with massive parallelisation to make efficient use of supercomputing resources to comute quantum mechanical properties. NWChem has been deployed on magnus to determine the atomic structures of amino acids which form the backbone of protein molecules. It is currently being deployed to evaluate polarisabilities of amino acids in order to facilitate the study of ion interactions with protein molecules and films.
FEniCS is a software tool that implements finite element methods, providing automated solvers of Poisson-Boltzmann models. FEniCS is readily available on Nimbus resources through Ubuntu cloud images and is employed to evaluate energies and forces in electrolytic systems. We are working with Pawsey staff to make FEniCS operational on magnus to enable computation of larger, more complex systems.
Pawsey resources also provide access to Lammps, molecular simulation software that enables investigation of larger scale structure than we can efficiently evaluate with quantum chemical software. We are investigating the applicability of Lammps for quantifying interactions of electrolyte ions with nonaqueous solvent molecules, which are used to obtain higher energy storage capacity than water.
Additionally, this project is being used to develop packaging for tools supporting computations which will be available to other groups through Ubuntu images on Nimbus.
List of Publications
Drew F. Parsons and Andrea Salis, A thermodynamic correction to the theory
of competitive chemisorption of ions at surface sites with nonelectrostatic physisorption. J. Chem. Phys. 151, 024701 (2019); DOI:10.1063/1.5096237
V. Esteso, S. Carretero-Palacios, P. Thiyam, H. Míguez, D. F. Parsons, I. Brevik, and M. Boström, Trapping of Gas Bubbles in Water at a Finite Distance below a
Water−Solid Interface, Langmuir 2019, 35, 4218−4223, DOI: 10.1021/acs.langmuir.8b04176
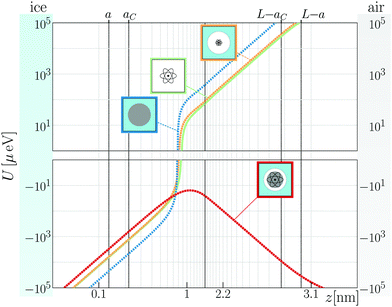
Mathias Boström, Robert W. Corkery, Eduardo R. A. Lima, Oleksandr I. Malyi, Stefan Y. Buhmann, Clas Persson, Iver Brevik, Drew F. Parsons, and Johannes Fiedler, Dispersion Forces Stabilize Ice Coatings at Certain Gas Hydrate
Interfaces That Prevent Water Wetting. ACS Earth Space Chem. 2019, 3, 1014−1022, DOI: 10.1021/acsearthspacechem.9b00019
Johannes Fiedler, Drew F. Parsons, Friedrich Anton Burger, Priyadarshini Thiyam, Michael Walter, I. Brevik, Clas Persson, Stefan Yoshi Buhmann and Mathias Boström, Impact of effective polarisability models on the near-field interaction of dissolved greenhouse gases at ice and air interfaces, Phys. Chem. Chem. Phys., 2019, 21, 21296–21304, DOI: 10.1039/c9cp03165k