Influence of N Doping on the Interfacial Lithiation behaviour of MoS2/Graphene Interface
The project aims to study the effect of N-doping on Li storage behaviours in MoS2/graphene hetero-structures during charging and discharging processes. Recently, two-dimensional MoS2/N-doped graphene (MoS2/NGr) composites have been a research focus in the field of energy materials. N-doped hetero-structures can remedy the large volume expansion and poor electronic conductivity of MoS2 electrode materials during cycles, which gives rise to a remarkable improvement on their capacity and cycling stability. However, the structural evolution of MoS2/NGr hybrid upon Li ion intercalation at the atomic level has not been fully explored. This project is designed to fully reveal the effect of N doping on the Li adsorption behaviour, thermodynamical stability and kinetic diffusion, as well as the mechanical properties of MoS2/Gr composites. Besides, K storage in hybrid will be studied in consideration of the excellent electrochemical performance of MoS2/NGr when it is used as anode material for potassium ion batteries. It is expected that the outcomes will provide a comprehensive understanding of Li storage mechanism in MoS2/NGr composites, and a guidance on developing MoS2/NGr electrodes in LIBs
Area of science
Energy Minerals
Systems used
Magnus
Applications used
The Challenge
Numerous efforts have been devoted to revealing the Li intercalation behaviour in MoS2/NGr hybrid, however, the rapid electrochemical reaction during charging and discharging processes makes it difficult for us to explore the evolution of structures and material properties during cycles with in-situ experiments. To reveal the micro-mechanism, this project is designed to simulate the Li storage behaviour in MoS2/NGr composites by using first-principles calculations.
The Solution
First, we compared the structural, electronic, mechanical properties of MoS2/NGr electrode materials before and after N-doping. Next, Li adsorption in hybrids was simulated, and the stable Li adsorption sites were identified. Here, the Li intercalation process was also unveiled. Then, we further studied Li kinetic diffusion in hybrids, including diffusion paths and energy barriers. Finally, it is shown that Li ions demonstrate a good adsorption capability in MoS2/NGr, with comparable kinetic diffusion capabilities to other typical two-dimensional electrode materials.
The Outcome
The main difficulties for this project are the huge computational tasks since the studies on Li adsorption and kinetic diffusion in hybrid involve a large number of simulations. The Pawsey Centre’s resources enable us to obtain the simulation results in a short time and ensure the smooth progress of simulations. Here, we would like to take this opportunity to thank the Pawsey Centre’s staff for their kind help and support.
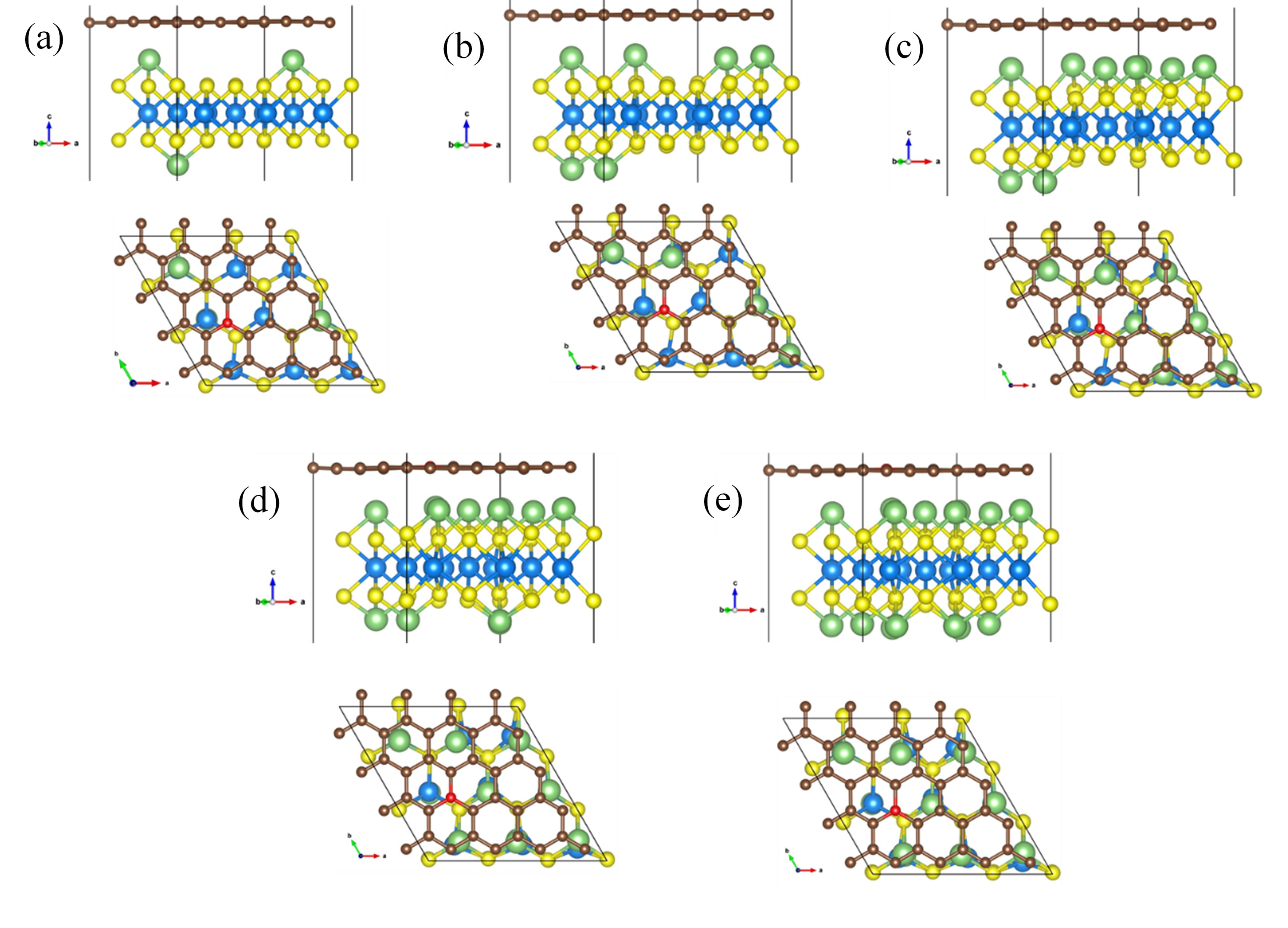
Stable 2H-MoS2/NGr interface structures with Li content of (a) 0.33, (b) 0.67, (c) 1.0, (d) 1.33 and (e) 1.67. Li, Mo, S and C atoms are presented by green, blue, yellow and brown, respectively.

The diffusion energy barriers and paths of isolated Li ion at (a, c) interface and on (b, d) MoS2 surface of 2H-MoS2/NGr and 1T-MoS2/NGr hybrids, respectively. Mo atoms refer to diffusion of Li-ion from one stable Mo top site (or the site near Mo) to its neighbouring stable adsorption sites (Mo2, Mo3 and Mo4).