Modelling the catalytic properties of water splitting on pure and defective gallium oxide surfaces
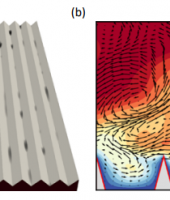
Research into the splitting of water by photocatalytic methods has risen in recent years to address the growing demand for sustainable energy. Gallium oxide has a wide range of uses, which includes UV detectors, semiconducting lasers and high temperature gas sensors. Due to its surface properties and wide band gap gallium oxide has been viewed as a potential candidate material for photocatalytic water splitting reactions. In this project we aim to investigate the catalytic properties of the surfaces of different polymorphs of gallium oxide. We aim to introduce defects and vacancies on the surface and determine if this enhances or diminishes the catalytic properties for splitting water. The overall outcome of the project will be to provide theoretical evidence of the types of phases and surfaces of gallium oxide that should be considered as the next generation of photocatalytic materials.
Area of science

Systems used
Magnus
Applications used
VASPThe Challenge
In this project we aim to investigate through the use of periodic Density Functional Theory (DFT) the catalytic properties of the low index surfaces of alpha and beta gallium oxide phases. Due to the sheer number of different adsorption sites, surfaces, phases and different water like species required to get a complete understanding of this process we need the use a supercomputer with a lot of processors for each simulation. Moreover, we require the use of non standard DFT (Hubbard U parameter, DFT-D and spin polarised calculations) to get a more realistic representation of the system.
The Solution
The DFT code we required, VASP, was compiled and optimised by staff in the Pawsey Centre. We were able to run all the calculations required to probe the reactivity and water splitting ability of a very important surface of gallium oxide. Moreover, we were able to probe the sensitivity and mobility of oxide capped GaN surfaces, which has direct relevance to the performance of important chemical sensors.
The Outcome
The time allocated on Magnus enabled us to explore the adsorption of different water like species on the surface of gallium oxide. Due to the number of different species and size of the system only a supercomputer could be used.
List of Publications
Theoretical study of the influence of surface effects on GaN-based chemical sensors, R. Anvari, D. Spagnoli, G. Parish, G. A. Umana-Membreno and B Nener, Applied Surface Science, 452, 75-86, 2018
Density Functional Theory simulations of water adsorption and activation on the (-201) β-Ga2O3 surface, R. Anvari, D. Spagnoli, G. Parish, and B Nener, Chemistry: A European Journal, 24, 29, 7445-7455, 2018
Effect of pH and structure on the chemical conductivity of AlGaN/GaN hererostructure based sensors, R. Anvari, D. Spagnoli, G. A. Umana-Membreno, G. Parish and B Nener, Sensors and Actuators B, 269, 54-61, 2018